Pictorial Review of Imaging Spectrum of Epilepsy on Magnetic Resonance Imaging
Abstract
Background: Objectives: The objective of our study is to review different imaging spectrums of epilepsy on MRI. To diagnose structural abnormalities in the brain associated with the cause of seizures with MRI. Subjects and Methods : It is a hospital-based prospective observational study that was carried out in 80 patients with a history of epilepsy in Narayana medical college and general hospital, Nellore in a period of 2 years from October 2019 to October 2021. All age group patients were included in the study without any gender predilection. Magnetic resonance imaging was performed on these patients and the results were recorded. Results: Out of these 80 patients, 30 patients (37.5%) had no abnormal neuroimaging findings. Most of the patients presented with generalized tonic-clonic seizures. The most common imaging findings were of cerebrovascular diseases (venous thrombosis/ infarct with gliosis) in about 16 cases (20%), developmental disorders in 17 cases (21.25%) of which mesial temporal sclerosis was the commonest (7 out of 17 cases), focal cortical dysplasia was seen in 3 cases. Other causes include infectious granulomas in 9 cases (11.25%) (tuberculomas and neurocysticercosis) and tumors in 2 cases (2.5%). vascular malformations (2 cases), and phakomatoses like tuberous sclerosis and Sturge weber syndrome (3 cases). Conclusion: MRI is a non-invasive modality with no radiation hazard and with its excellent gray-white matter resolution and multiplanar imaging capability helps in the detection of small and subtle lesions. With appropriate imaging protocol, our study observed that MR evaluation with increases sensitivity and specificity in the evaluation of seizures.
Keywords
Epilepsy, MRI
Introduction
Seizure is a most common medical disorder and neuroimaging plays a valuable role in the evaluation of etiological factors for seizures. Epilepsy is a disorder of the brain characterized by two or more seizures unprovoked by any immediately identifiable cause. It is the most common neurological disorder in developing countries.[1]
An epileptic seizure is a result of abnormal, excessive neuronal activity arising in the grey matter of the cerebral cortex.[2]
Magnetic resonance imaging plays an important role in the diagnosis of any neurological conditions that lead to epilepsy.
MR imaging because of its excellent soft-tissue contrast, acts as a most valuable tool for preoperative evaluation, allowing for a detailed evaluation of anatomy to localize epileptogenic focus. When compared to CT, MRI is free from beam hardening artifacts.[3]
MRI protocol routinely includes T1 and T2 weighted images, fluid-attenuated inversion recovery sequence (FLAIR). MR Images should be obtained in at least two orthogonal planes covering the whole brain with minimum slice thickness not exceeding 3mm. In suspected cases of temporal lobe epilepsy, oblique coronal plane images obtained perpendicular to the long axis of the hippocampus are the best.[4]
Aims and objectives
-
The objective of our study is to review different imaging spectrums of epilepsy on MRI.
-
To diagnose structural abnormalities in the brain that are associated with the cause of seizures with MRI.
Subjects and Methods
Various Causes of Epilepsy
Perinatal hypoxia and ischemia, intraparenchymal bleeds, trauma, and acute CNS infections, metabolic disorders, developmental and genetic disorders are the causes of seizures in newborns.
Febrile seizures, CNS infections, trauma, idiopathic, developmental, and genetic disorders in infants and children.
Illicit drug abuse can be one of the factors in adolescent age groups and young adults.
Cerebrovascular disease, brain tumors, metabolic causes, alcohol withdrawal, Alzheimer’s, and degenerative disease are causes of seizures in older adults(>35yrs).[5]
Mesial temporal sclerosis
It is the most common cause of epilepsy in the adolescent age group. The mesial temporal lobe includes the hippocampus, amygdala, and parahippocampal gyrus. In cases of medically intractable temporal lobe epilepsy, the most common etiology identified is hippocampal sclerosis. Loss of neurons and gliotic changes lead to reorganization of the neuronal circuit within the hippocampus which consequently results in an epileptogenic focus.[6]
Primary signs include the loss of bulk of hippocampus, hyperintensity on both T2W and FLAIR images, and loss of hippocampal architecture and normal digitations of the head.
Secondary signs include atrophy of the mamillary body, fornix columns, amygdala, collateral white matter bundle, and dilatation of the temporal horn.[7]

Developmental disorders
Cortical developmental anamolies are more commonly identified as causes of epilepsy and neurodevelopmental deficits and are best demonstrated with MRI. Neuroimaging Commission of the International League against Epilepsy, subdivide malformations as follows.[8]
-
Diffuse cortical malformations which include the lissencephaly pachygyria spectrum which in turn includes agyria, pachygyria, lissencephaly, Polymicrogyria, microcephaly, megalencephaly, and microdysgenesis.
-
Focal or multifocal cortical malformations: focal cortical dysplasia, hemimegalencephaly, focal polymicrogyria, tuberous sclerosis,
-
Heterotopias.
Focal cortical dysplasia is one of the most common causes of epilepsy, resulting in focal cerebral dysgenesis in the temporal lobes. Cortical thickening on at least 3 or more continuous slices, as well as loss of the gray-white matter junction, are MRI findings in patients with FCD.
An aberrant signal intensity tapering in a linear, curved, or funnel-shaped pattern stretching from the cortical white matter junction to the ependymal surface of the lateral ventricle.[9]

Polymicrogyria: It is a malformation of the deeper layers cortex in which multiple tiny gyri with abnormal sulcation are noted. It may be unilateral or bilateral. It is characterized by multiple small gyri with a predilection for sylvian fissure.[9]

Lissencephaly
Lissencephaly pachygyria complex can be further divided into two types.[9]
Type I (Classic) can be seen as a poorly formed gyri and smooth outer surface, commonly associated with band heterotopia.
Type II (Cobblestone) has a microlobulated pebbly cortical surface with a reduction in normal sulcation.
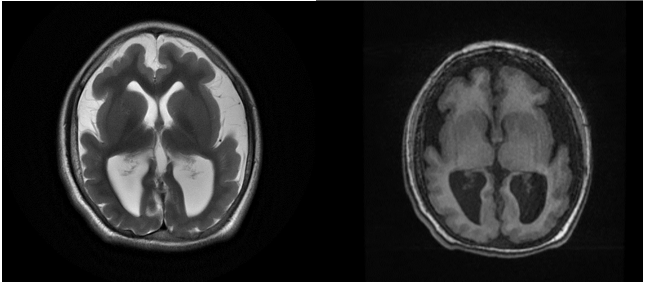
Heterotopias
These include a group of conditions characterized by interruption of normal neuronal migration.
These can be divided into nodular heterotopias and diffuse heterotopias. On MRI heterotopic tissue follows the signal of grey matter in all sequences.[9]

Vascular Causes
Cerebrovascular stroke is a commonest cause of epilepsy in elderly patients.
Hemorrhagic stroke or venous thrombosis leading to stroke are commonly associated with post-stroke seizures. Hemorrhage within brain parenchyma leads to seizures due to irritation by blood products. Late-onset seizures are seen due to gliotic scarring of infarcted cortical tissue.[10]

Seizures are noted in about 40-45% of patients with venous thrombosis leading to venous infarct and hemorrhage.
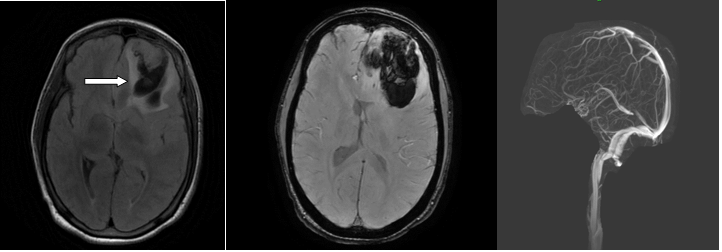
Vascular malformations
The most common vascular malformations causing seizures in epilepsy patients include arteriovenous malformations and cavernous malformations. Presenting symptoms of these conditions include headache, seizures, focal neurological deficits.
Arteriovenous Malformations (AVM)
These are congenital, developmental anomalies of blood vessels wherein a tangle of blood vessels is noted with no intervening capillary network and with direct arteriovenous shunting of blood.[11]
Steal phenomena due to arteriovenous shunting may result in cerebral ischemia. Gliosis and hemosiderin deposition from hemorrhage in the brain parenchyma are the possible mechanisms of seizures.

Cavernous malformations
Cavernomas are well-defined vascular spaces that contain blood in various stages of evolution. No intervening neural tissue is seen within the lesion.
Characteristic MR appearance is mulberry-like heterogeneous hyperintense signal centrally on all pulse sequences, surrounded by a hemosiderin rim which is of low signal intensity. Because hemosiderin results in magnetic susceptibility artifacts, gradient-echo images will tend to be more useful for detecting small cavernomas.[11]

Infections:
Neurocysticercosis
Neurocysticercosis (NCC) of the central nervous system is a clinical condition in which CNS infestation occurs by the larval stage of the pork tapeworm Taenia solium.
Involvement can be parenchymal. Involvement of ventricles, subarachnoid spaces, or a combined involvement of all these sites can be seen. Inflammatory mediators that are released in response to the dead and degenerating parasite result in seizures.
Clinical manifestations depend on the number, size, localization, viability, and different stages of cyst degeneration.
Lesions on MRI can be of various stages depending on the evolution of the larva, lesions in different stages of the disease can be seen simultaneously. Four different stages are the following.[12]
The vesicular stage: In this stage, thin-walled cystic lesion with intracystic fluid showing signal intensity similar to that of CSF in all sequences. No host reaction is seen. FLAIR images show the scolex as an eccentrically punctuate bright signal intensity within the cysts.
In the colloidal stage, inflammatory changes develop around the cysticercus, its wall thickness, and the vesicular fluid takes on a gelatinous colloidal aspect. Contrast enhancement is usually peripheral, and perilesional edema is seen. Slightly hyperintense on T1W, T2WI and FLAIR images.
Granular nodular stage, Dead and retracted parasite in the lesion begins to mineralize. There is nodular or ring enhancement, with less edema.
In the nodular calcified stage, the lesion is completely mineralized. Calcified cyst remnant seen with no edema.
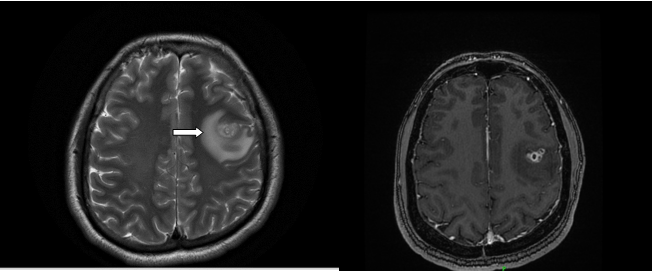
Tuberculomas
Tubercular infection of the central nervous system results in meningitis and tuberculoma formation.
Risk factors include elderly age, low economic status, and immunosuppressive conditions like human immunodeficiency virus (HIV), and lymphoma.
Leptomeningitis is common and can cause hydrocephalus, neuropathies, and sometimes deep gray matter infarction.
Single or multiple intraparenchymal tuberculomas may be seen. Tuberculomas can be either non-caseating or caseating with a solid center or caseating with a liquid center. The non-caseating granuloma shows homogeneous contrast enhancement. The caseating solid granulomas appear as ring-enhancing lesions with vasogenic edema.
A characteristic MR spectral pattern that primarily involves long-chain lipids is seen in tuberculomas. MR imaging along with spectroscopy increases the specificity of diagnosis of intracranial tuberculomas.[7]

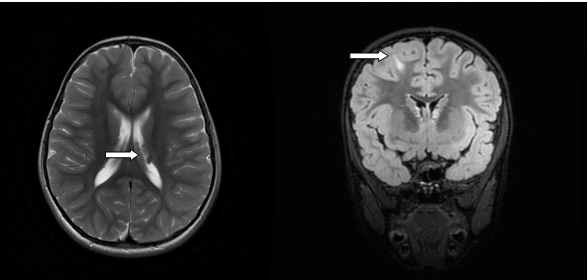
Tuberous Sclerosis:
Tuberous sclerosis is an autosomal dominant disorder that results in hamartomas in multiple organs. It is an inherited disorder with a classic triad of adenoma sebaceum, epilepsy, and mental retardation. The cortical hamartomas are called tubers and are similar to cortical dysplasia.
Subependymal nodules are small lesions protruding into the lateral ventricles. Some of these nodules may be calcified.[13]
Tubers are regions of T1 iso/hypointensity and T2 hyperintensity in subcortical regions.

Other causes include metabolic disorders which are not included in this study.
|
MR diagnosis |
No. of patients |
Percentage |
|
Normal study |
30 |
37.5 |
|
Cerebrovascular causes (Infarct with gliosis and venous thrombosis) |
16 |
20.0 |
|
Developmental malformations |
10 |
12.5 |
|
Mesial temporal sclerosis |
07 |
08.75 |
|
NCC |
05 |
06.25 |
|
Tuberculoma |
04 |
05.00 |
|
Low grade Glioma |
02 |
02.5 |
|
Cavernoma |
02 |
02.5 |
|
Tuberous sclerosis |
02 |
02.5 |
|
Cerebral abscess |
01 |
1.25 |
|
Sturge-weber syndrome |
01 |
1.25 |
|
Total |
80 |
|

Tumors
Neoplasms associated with epilepsy are commonly located in the cortex and are not usually associated with mass effect or vasogenic edema. The temporal lobe is most common site.
These include: –
1. Low-grade astrocytomas.
2. Oligodendroglioma.
3. Ganglioglioma.
4. Dysembryoblastic neuroepithelial tumor (DNET), and
5. A pleomorphic xanthoastrocytoma (PXA).
Low-grade astrocytomas, fibrillary subtype are usually ill-defined, infiltrative tumors that usually do not enhance with gadolinium whereas pilocytic astrocytomas are well-defined tumors with a mural nodule that shows post gadolinium enhancement.
Oligodendrogliomas are usually located in cortical and subcortical regions and with gyriform calcifications with variable contrast enhancement and adjacent bony changes of the calvarium. They are common in the frontal or temporal lobe.
Gangliogliomas are cortical-based solid cystic lesions with variable contrast enhancement and no significant mass effect.[11] Calcification is often present Dysembryoplastic neuroepithelial tumors (DNET) are benign, cortical-based tumors of low- grade. They are multicystic, and multinodular lesions with variable contrast enhancement. They are primarily seen in children and young adults. Bony remodeling may be seen.
Pleomorphic xanthoastrocytomas (PXAs) are superficially located solid cystic masses with large cystic component, enhancing solid component and mural nodule. They are located adjacent to the leptomeninges and involve them.[11]
Leptomeningeal involvement is characteristic of this tumor. The solid component usually enhances and is calcified about half of the cases.
Conclusion
Seizure disorder is one of the common presenting problem in neurological practice and MRI brain plays a crucial role in the evaluation and proper diagnosis of these patients.
Treatment to the patient can be effectively given when the diagnosis is made accurate. The Sensitivity and specificity of MRI is high in detecting the causative of seizures. MRI has the added advantage of being a nonionizing modality. MRI has an excellent spatial resolution, good soft-tissue contrast, and has the ability to create multiplanar imaging. With these features, it became the modality of choice in the evaluation of patients with seizures. Using advanced MRI sequences such as ASL, MRS, functional MRI etc., we can further categorize and localize the lesion for specifically targeted management.[14,15]
MRI brain identifies epileptogenic substrates accurately thus it helps in determining the treatment plan and the prognosis can also be evaluated further. Follow-up scans help to see any residual/recurrent lesions which may cause epilepsy in the future. Our study showed the role of MRI in epilepsy in early and proper diagnosis and proved MRI has high sensitivity and specificity.